 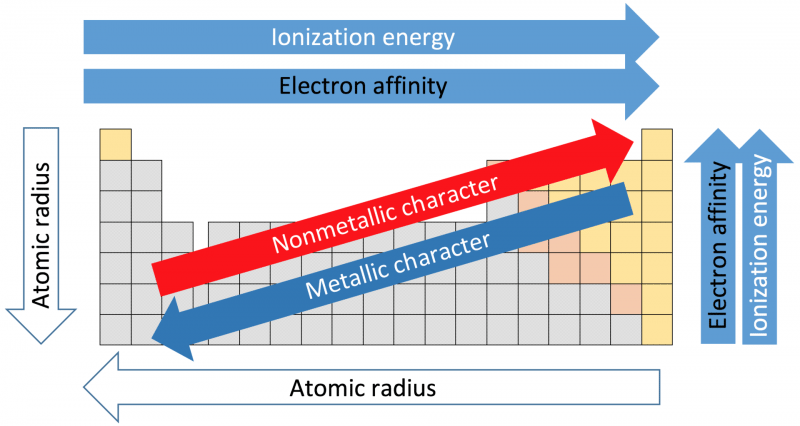
I am not sure of how correct my knowledge is. This is the way I personally see it at least. So they would still be more active going to the left-hand side of the table. In Metals: Period - reactivity decreases as you go from left to right. The Group Number of an element is the same as the number of electrons in its outer shell. Second, assuming they could do both (lose and gain) it would still be far easier to lose 2 or 3 electrons than add like 6 more to fill the valance shell. Group - reactivity decreases as you go down the group. Step 1: Identify the Group Number of the given element in the periodic table.  
Now intuitively it could be thought that an atom would like to have an octet of valance electrons and since most of the nonmetals are on the right-hand side of the table, the closest "octet" would be to the right, achieved by gaining electrons, and the closer it is to the right, the more it wants that electron (becomes more active like halogens).īut for metals it's the very opposite, they want to LOSE electrons because Nonmetals can do both, lose and gain electrons. Well first off metals can ONLY lose electrons, so they will always either gain a positive charge or will have a positive oxidation state. reaction with oxygen is slow at room temperature, but they can ignite around 150-200 ☌. A screenshot is preferable to a picture of your laptop screen. The Lanthanides have the following reactions: oxidize rapidly in moist air. By following the trend summary in the figure below. This is indicated by their low electronegativities. Within a compound, metal atoms have a relatively low attraction for electrons. Metals tend to lose electrons in chemical reactions, as indicated by their low ionization energies. Please do not ask for help acquiring, preparing, or handling illicit substances or for help with any activity that does not fall within the confines of whatever laws apply to your particular location.īonus points: If submitting a picture please make sure that it is clear. Metallic character refers to the level of reactivity of a metal. Any infractions will be met with a temporary ban at the first instance and a permanent ban if there is another. It is also important that you describe the specific part of the problem you are struggling with. The reactivity of metals increases further left along a period, and further down a group. It is OK if you are a little (or a lot!) stuck, we just want to see that you have made an effort. Reactivity is dependent on the classification of an element (metals and non-metals), as they both have differing periodic trends. Please complete any questions as much as you can before posting. We will not do your homework for you, so don't ask. Please flair yourself and read over the rules below before posting. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
